MACUSTAR study participants
The MACUSTAR study (ClinicalTrials.gov Identifier: NCT03349801), a prospective multicenter, low-intervention natural history study, aims to identify novel biomarkers for iAMD44. The study’s design and participant selection criteria have been outlined in prior publications44,45,46. Enrolment occurred from March 2018 to February 2020, selecting one eye per participant for the study, prioritizing the eye with better visual acuity when both eyes met the inclusion criteria. In accordance with the Declaration of Helsinki, ethical guidelines were strictly followed, with informed written consent obtained from all participants. The study includes four groups: early AMD, iAMD, late-stage AMD, and a control group3. Consistent with the classification system proposed by Ferris et al., iAMD was defined by the presence of large sub-RPE drusen (> 125 µm) and/or any AMD-related pigmentary abnormalities in both eyes3. If the fellow eye had an extrafoveal GA lesion, it was required to be no larger than 1.25 mm2. Early AMD was characterized by medium-sized drusen (63–125 µm), while late-stage AMD included MNV and/or central GA cases. All image grading and classification of study eyes were performed centrally at the GRADE Reading Center Bonn by trained and independent graders, following the MACUSTAR standardized grading protocol44.
In total 301 participants were recruited and included in the cross-sectional part of the MACUSTAR study (early AMD n = 34, iAMD n = 168, late AMD n = 43, controls n = 56). Out of those, 26 participants were not assessed in this study due to incomplete functional data (no AMD n = 2, iAMD n = 8, late AMD n = 6) or missing determination of the rEZR at positions of the MAIA stimulus grid (no AMD n = 1, iAMD n = 8, late AMD n = 1). Reasons for missing microperimetry assessments were mostly procedural errors, e.g. incorrect grid use or incomplete upload of data.
Imaging protocol
Participants underwent multimodal retinal imaging following standardized operating procedures conducted by certified study site personnel. Prior to imaging, pupil dilation was achieved using tropicamide 0.5% and phenylephrine 2.5% eye drops. Retinal imaging included combined confocal scanning laser ophthalmoscopy for near-infrared reflectance imaging [Automated Real-Time mode (ART) ≥ 30 single frames] and SD-OCT [30° × 25°, enhanced-depth-imaging, high-speed mode, 241 B-scans, distance 30 µm, ART mode = 9] which was acquired using the Spectralis HRA + OCT device (Heidelberg Engineering, Heidelberg, Germany).
Determination of the relative EZ reflectivity
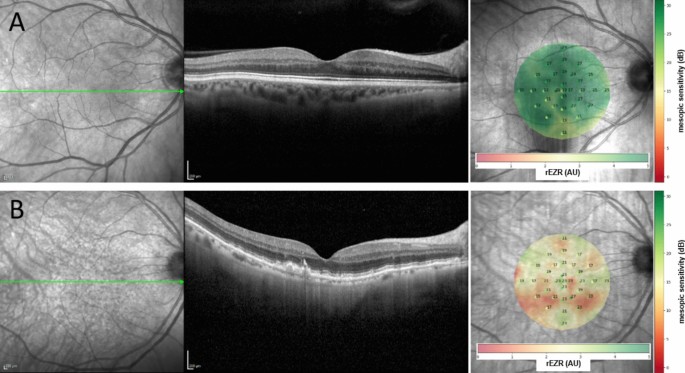
The rEZR was calculated as the ratio of the peak reflectivity of the EZ to the peak reflectivity of the ELM, using raw OCT images to ensure precise analysis of native, untransformed reflectivity signals (dynamic range: 0 to 1 [arbitrary units, AU]). This ratio minimizes the impact of acquisition-related variability or noise, including potential differences in illumination or scan intensity, as such effects are expected to influence both layers similarly, effectively canceling out when expressed as a ratio12,47. The ELM was selected as the reference layer due to its well-documented stability across a wide range of retinal eccentricities, including the fovea, as reported in previous studies6,48. Furthermore, as a non-neural structure, the ELM is less susceptible to reflectivity changes associated with aging or early retinal degeneration, making it a reliable benchmark for rEZR determination.
An automated algorithm (Python Software Foundation, Python Language Reference, version 3.9. Available at [ annotated code available at: bisselma/relEZIquantification) was employed to calculate the rEZR, as previously validated and described in detail15,47,49,50. Briefly, segmentation coordinates, obtained via a deep learning-based approach, were superimposed on the raw, non-logarithmic OCT images and used to straighten each B-scan along the RPE51. This alignment ensured accurate rEZR calculation, even in eyes with pronounced posterior pole curvature. Regions of interest were defined at adjoining nine-pixel intervals along the x-axis of each B-scan. Within these regions of interest, reflectivity profiles were generated and the EZ and ELM peak reflectivities were automatically identified. Predefined subregions, based on the 95% prediction interval of reflectivity profiles, were used to reliably detect the EZ and ELM peaks (Fig. 2)47. This process facilitated both global and spatially resolved rEZR calculations across each B-scan (n = 241) of the SD-OCT raster scan.
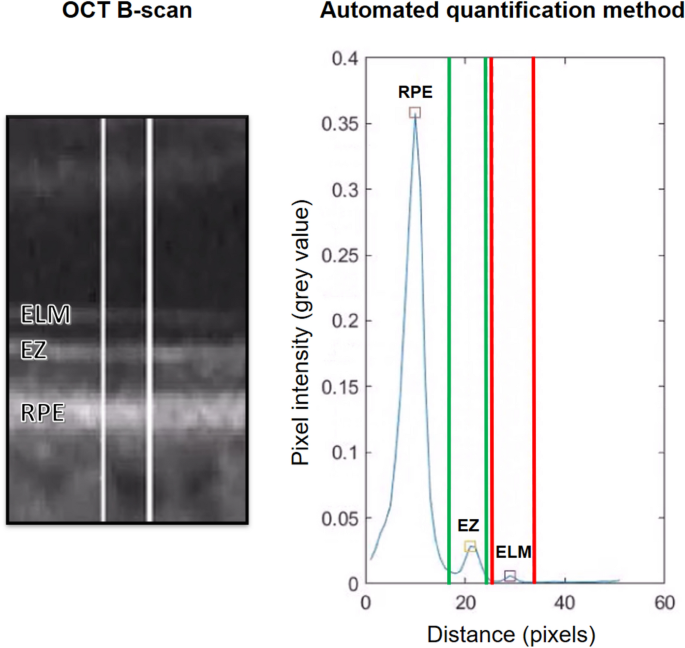
A representative case illustrating peak determination by the automated method. The left panel shows an OCT B-scan with the external limiting membrane (ELM), ellipsoid zone (EZ), and retinal pigment epithelium (RPE) labeled. White lines indicate the region of interest used for reflectivity analysis. The right panel presents the corresponding pixel intensity profile, where green and red vertical lines mark the peak detection areas for EZ and ELM, respectively. Colored rectangles denote peaks identified by the algorithm: RPE (red), EZ (yellow), and ELM (purple).
To reduce interference from structural changes, retinal areas affected by sub-RPE drusen were automatically excluded from further analysis. These regions were identified as areas where the separation between the RPE and Bruch’s membrane exceeded 15 pixels (~ 100 µm on the image y-axis in high-speed Spectralis OCT imaging). Additionally, regions with MNV or GA, where the absence of EZ or ELM peaks impedes accurate reflectivity assessment, were also excluded.
Functional probing
As part of the MACUSTAR protocol, BCVA, low-luminance visual acuity, Moorfields Acuity Test, and low-luminance deficit (calculated as low-luminance visual acuity minus BCVA) were assessed using the ETDRS charts and quantified on a logMAR scale. The MAT utilizes pseudo high-pass letter optotypes, designed to reduce low spatial frequency cues, making optotype recognition more dependent on resolution. This improves repeatability and may enhance sensitivity to early AMD-related vision loss52. Additionally, contrast sensitivity was assessed using the Pelli Robson Contrast Sensitivity Test, measured on the logCS scale. The assessment of retinal function further comprised mesopic FCP utilizing the MAIA microperimeter (software version 2.5.1, iCare, Padua, Italy). Functional testing procedures have been extensively described in previous publications23,29.
Specifically, with regard to FCP, a customized testing grid consisting of 33 stimulus points located at fixed degrees (0°, 1°, 3°, 5°, and 7°), with the fovea serving as the central point, was employed53. Mesopic FCP employed a Goldmann size III stimulus (0.43° diameter) for 200 ms, with a dynamic range of 36 dB. A 4–2 strategy adjusted stimulus intensity based on responses to determine thresholds, with a background luminance of 1.27 cd/m2. A fixation target (3° radius, 1-pixel thickness) aided in stable fixation. After dilation with 1% tropicamide, participants underwent 5 min of dark adaptation in a fully dark room to ensure consistent testing conditions.
Analyses of the rEZR and retinal function
Analyses of the rEZR and retinal function employed global, locally averaged, and spatially resolved models. In the global model, the mean rEZR from the entire SD-OCT scan was tested for association with the mesAT in decibels across all 33 stimulus points.
In the locally averaged model, the rEZR was determined at the 33 stimulus points of the FCP grid, assessing areas twice the diameter of each point. The mean rEZR per participant was then tested for its association with the average threshold.
The spatially resolved model calculated the rEZR at each FCP point (twice the stimulus point diameter) to test associations between the rEZR and localized retinal sensitivity (dB), providing a more detailed analysis of structural and functional associations.
The associations between the rEZR and other functional tests were also tested globally and spatially. The global model used the mean rEZR for the entire SD-OCT scan, while the spatially resolved analysis focused on the central subfield (1 mm diameter) of the ETDRS grid to test associations with functional test values.
Alignment of structural and FCP-derived functional study data
For a spatially-resolved analysis of the rEZR’s functional impact, structural and functional study data were precisely aligned using corresponding en-face near-infrared reflectance images from FCP testing and confocal scanning laser ophthalmoscopy imaging of the SD-OCT dataset (Fig. 3). The initial pre-processing included cropping and resizing the FCP-derived near-infrared reflectance images to match the frame (30° × 30°) and size (768 × 768 pixels) of the confocal scanning laser ophthalmoscopy near-infrared reflectance images. The alignment of the structural and functional data was then accomplished using the “SuperRetina” registration method, a deep learning-based technique specifically trained for the accurate registration of retinal imaging data54. This approach allows for a reliable alignment, capable of compensating for noise, artifacts, or variable image quality. The alignment of deep learning-registered images was visually verified by overlaying them in two color channels to ensure accuracy. No deviations were observed, confirming the images were consistently aligned and suitable for analysis.
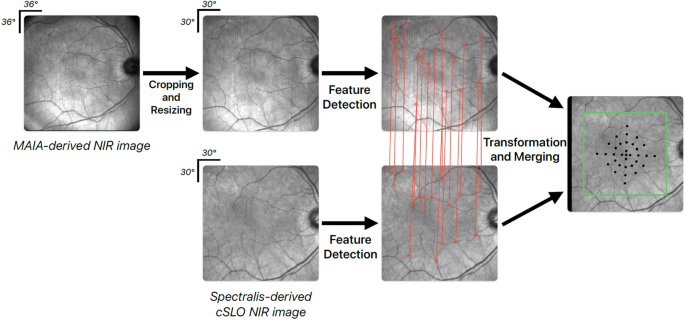
Illustratration of the alignment process of structural, i.e. Spectralis spectral-domain optical coherence tomography (SD-OCT) imaging derived, and functional, i.e. MAIA fundus-controlled perimetry (FCP)-derived, study data. The near-infrared image (NIR) of the FCP-data was cropped and resized to match the image dimensions of the SD-OCT derived NIR image. The deep-learning algorithm “SuperRetina” by Liu et al. was applied to align the NIR images of the FCP and SD-OCT study data using landmark correspondences54.
Statistical analysis
The study included a descriptive analysis that summarized the baseline characteristics of all participants with valid MAIA exams and available rEZR, focusing on the means and standard deviations.
Analyses were performed using multivariable linear models, adjusted for age, sex, and AMD stage, with functional measures as dependent variables. Additionally, univariate linear regressions were conducted to individually assess these relationships. For the spatially resolved FCP-analysis, a linear mixed-effects model was applied, using the retinal sensitivity values (dB) at each topographically aligned stimulus point as the outcome measures. A patient’s random intercept term was included to account for multiple measurements within the same eye. Further, the spatially resolved model included a spline term for the eccentricity of the rEZR within the volumetric SD-OCT raster scan. For all models, coefficient estimates were assessed, including calculation of 95% confidence intervals.
Descriptive p-values are reported without adjustment for multiple testing, as each functional parameter was analyzed independently for its association with the rEZR in this exploratory study. Since the analyses were not conducted within a single multivariate framework, the risk of false positive findings is minimized, and statistical adjustment for multiple comparisons is not required. A significance level of 0.05 was considered. All analyses were performed using R Version 4.3.055.
link